The Biochemistry Section currently focuses on discovering the functions and interrelationships among proteins mutated in familial Parkinson’s disease and ALS. This has led the unit to explore the molecular mechanisms of autophagy, mitochondrial dynamics, apoptosis and mitochondrial quality control.
Using biochemical, cell biological and animal studies we have found that the products of two genes mutated in autosomal recessive forms of Parkinson’s disease, PINK1 and Parkin, normally work together in the same pathway to govern mitochondrial quality control, bolstering previous evidence that mitochondrial damage is involved in Parkinson’s disease. PINK1 is a kinase located on mitochondria, and Parkin is an E3 ubiquitin ligase normally located in the cytosol. When mitochondria are damaged, PINK1 accumulates on the outer mitochondrial membrane where it phosphorylates ubiquitin chains. These phosphorylated ubiquitin chains on the outer mitochondrial membrane bind to cytosolic Parkin and activate Parkin’s E3 ubiquitin ligase activity yielding a feedback amplification loop that drives mitophagy to completion. These finding reveal a cell biology pathway where Pink1 function is paired with that of Parkin supporting the model that mutations that inactivate this quality control pathway leads to parkinsonism.
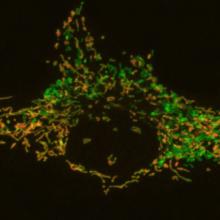
Downstream of PINK1/Parkin the machinery that mediates autophagosome recognition of damaged mitochondria links this pathway to genes mutated in ALS. Using CRISPR/Cas9 to knock out a series of autophagy receptors reveals their function and hierarchy in autophagosome engulfment of mitochondria. For example, Optineurin and NDP52 bind to ubiquitin chains on mitochondria and also recruit autophagy machinery proteins, including the upstream kinase Ulk1 and the downstream autophagosome marker, LC3, to induce autophagosome engulfment of the damaged mitochondria. Animal models that display mitochondrial damage require endogenous Parkin to prevent domaminergic neuron loss and movement disorders. Interestingly, in a murine model of mitochondrial damage, the product of the kinase PINK1 (phospho-S65 ubiquitin) increases several fold in the cortex, representing a biomarker of PINK1 activity. We are further examining biomarkers of PINK1/Parkin activity in vivo to define roles of mitochondrial quality control in a series of physiological and pathological states.
