The National Institutes of Health (NIH) and the U.S. Department of Veterans Affairs (VA) are working on a pioneering collaborative research project, the VA-NIH Investigative Deep Phenotyping (IN-DEPTH) of Gulf War Veterans Health.
Gulf War Illness
The National Institutes of Health
The National Institutes of Health (NIH) is a part of the U.S. Department of Health and Human Services and is the nation's medical research agency. It is made up of 27 Institutes and Centers, each with its own specific research area. NIH is unlike other hospitals because its sole purpose is to conduct biomedical and public health research studies. Only patients under research protocols are evaluated at the NIH.

Useful Resources
- Virtual Tour of the NIH Clinical Center - YouTube
- NIH Clinical Center: A Guide for Patients Entering Campus - YouTube
- NIH Clinical Center: Patients are our Partners - YouTube
Project Overview
Project IN-DEPTH is two studies: VA IN-DEPTH and NIH IN-DEPTH

The project involves two separate research studies –VA IN-DEPTH and NIH IN-DEPTH, which work together for the common goal of learning more about GWI.
The VA IN-DEPTH study identifies Gulf War Veterans with GWI and healthy veterans that served in the war.
The NIH IN-DEPTH study is focused on making scientific measurements of the Gulf War veterans identified during VA IN-DEPTH. The goal of the NIH IN-DEPTH research study is to thoroughly describe the clinical and biological characteristics of GWI. Participants in the study will not receive treatment. However, insights from this study may lead to:
- More definitive diagnostic criteria
- Development of new diagnostic testing
- Identify potential treatments
All Participants are Gulf War Veterans
All participants must be 48-70 years old and have at least a 7th grade education. In addition, all eligible participants must fit into one of the two groups below:
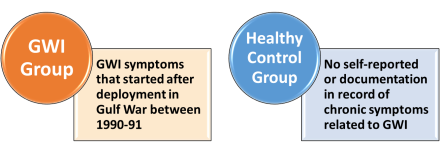
- Inpatient Gulf War illness group: Those with GWI symptoms that started after deployment to the Gulf War between 1990 and 1991.
- Inpatient Healthy Veteran Control group: No self-reported or documentation in record of chronic symptoms related to GWI.
Study Flow
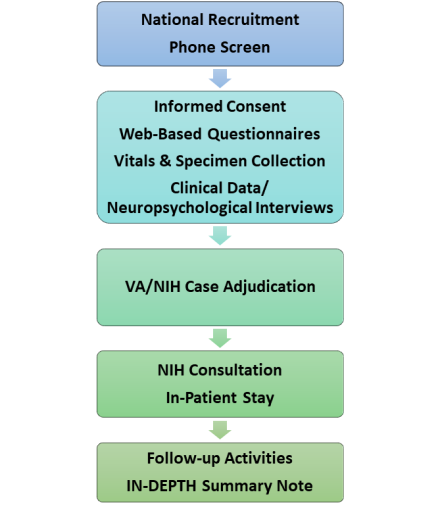
The IN-DEPTH study has five main parts. It starts with the VA IN-DEPTH study of recruitment and screening. It is followed by the NIH IN-DEPTH study, which focuses on collecting and making scientific measurements and information. Each individual will receive an IN-DEPTH summary note and be offered the option to participate in other study follow-up activities.
What happens during NIH IN-DEPTH?
Comprehensive testing will be done during a 14-day hospital admission at the NIH Clinical Center to look at multiple body systems affected by Gulf War Illness and how these systems operate functionally and structurally at rest as well as when the body is placed under stressors. Additional study measures will be done at home, such as wearing an activity monitor and completing a diary of activity and responses.
Interested individuals for the NIH study will initially complete a telephone pre-screening with the NIH study staff. The NIH study team will receive medical records regarding your diagnosis and receive permission to speak to health care providers. After review of the records, you will be contacted to coordinate the visit to the NIH Clinical Center.
Travel arrangements to and from the Clinical Center will be coordinated with the patient and the study team. After arriving at the hospital, you will be assigned a hospital room. Most patients will stay in the Clinical Center, which has many different staff members available to help throughout the visit. During your stay, members of your study team will be administering tests and assisting you with the various activities as well as escorting you to different departments of the Clinical Center required to complete the study testing.
Testing during NIH IN-DEPTH
Below is a list of optional and required procedures.
Required Procedures
- An explanation of the study and signing of the research study consent form
- Medical history
- Physical exam and strength testing
- Saliva, cheek swab, and stool sample collection
- Questions about the participant’s life and their quality of life
- Questions about the participant’s mental health
- Tests of sweating. breathing, blood pressure in different positions, and heart rate
- Questions about the participant’s diet and food preferences
- Interviews about fatigue, symptoms, diet, health, and mood
- Thinking and memory tests
- Wearing an activity monitor and completing a fatigue diary
- Blood collection
- Urine collection
- A lumbar puncture (spinal tap)
- Heart (Holter) monitor
- An exercise stress test. Participants will pedal a stationary bike until too tired to continue.
- Sleeping in a metabolic chamber- a room that monitors energy and nutrient balance.
- Eat a special metabolic diet where all of the food is weighed before being prepared
- Wear a transparent hood to measure energy use
- X rays to look at body composition
- Echo
- EKG
- PFT
- Chest CT
Optional Procedures
- CT Cerebrum
- Autonomic Testing
- Functional MRI
- Brain transcranial magnetic stimulation.
- Magnetic resonance imaging (MRI) of the brain.
- Muscle and skin biopsies
- Participants who are on medications that could interfere with the study testing may be asked to stop those medications prior to study procedures, if it is possible to do so safely.
How to Volunteer for Project IN-DEPTH
The goal of the VA IN-DEPTH study is to identify participants for eligibility in the NIH study. Interested veterans can volunteer to participate in Project IN-DEPTH by contacting the VA-IN DEPTH study team, who are actively recruiting participants nationwide. After an initial phone screening process and study consent is completed, the eligibility screening is performed. It includes medical tests, questionnaires, and interviews to better understand a veteran’s health and service during the Gulf War. This will be done primarily via phone and video calls.
There is also a home visit to collect patient vital signs, such as blood pressure, and blood samples. There are no costs for this screening; participants will receive a stipend for the activities completed. After completing the evaluation, the adjudication committee, comprised of investigators from the VA and NIH, meet to review the details of the evaluation to determine who meets the eligibility criteria for participating in NIH IN-DEPTH.
Interested veterans are encouraged to contact the VA IN-DEPTH study staff. For more information on the VA IN-DEPTH protocol including how to participate, please refer to Project IN-DEPTH - War Related Illness and Injury Study Center (va.gov).
Contact the VA IN-DEPTH Team: vhawas.indepth@va.gov
Contact the NIH IN-DEPTH Team: gwiindepth@ninds.nih.gov
Frequently Asked Questions
A comprehensive list of FAQs can be found here.
